Fluorine: The Chemical Predator. When One Extra Proton Turns an Element into a Monster. Part 9
“The most dangerous predators are not the biggest, but the hungriest.”
— Folk wisdom
We have journeyed from the symmetrical Carbon through the asymmetrical Nitrogen to the powerful oxidizer Oxygen. Now we approach the pinnacle of chemical aggression — Fluorine.
It is not just a “very active element”. It is a substance that eats through glass, explodes on contact with water, and forms compounds with almost all elements of the periodic table. And we will explain why — simply by looking at the architecture of its nucleus.
📐 Engineering Analysis of the Nucleus
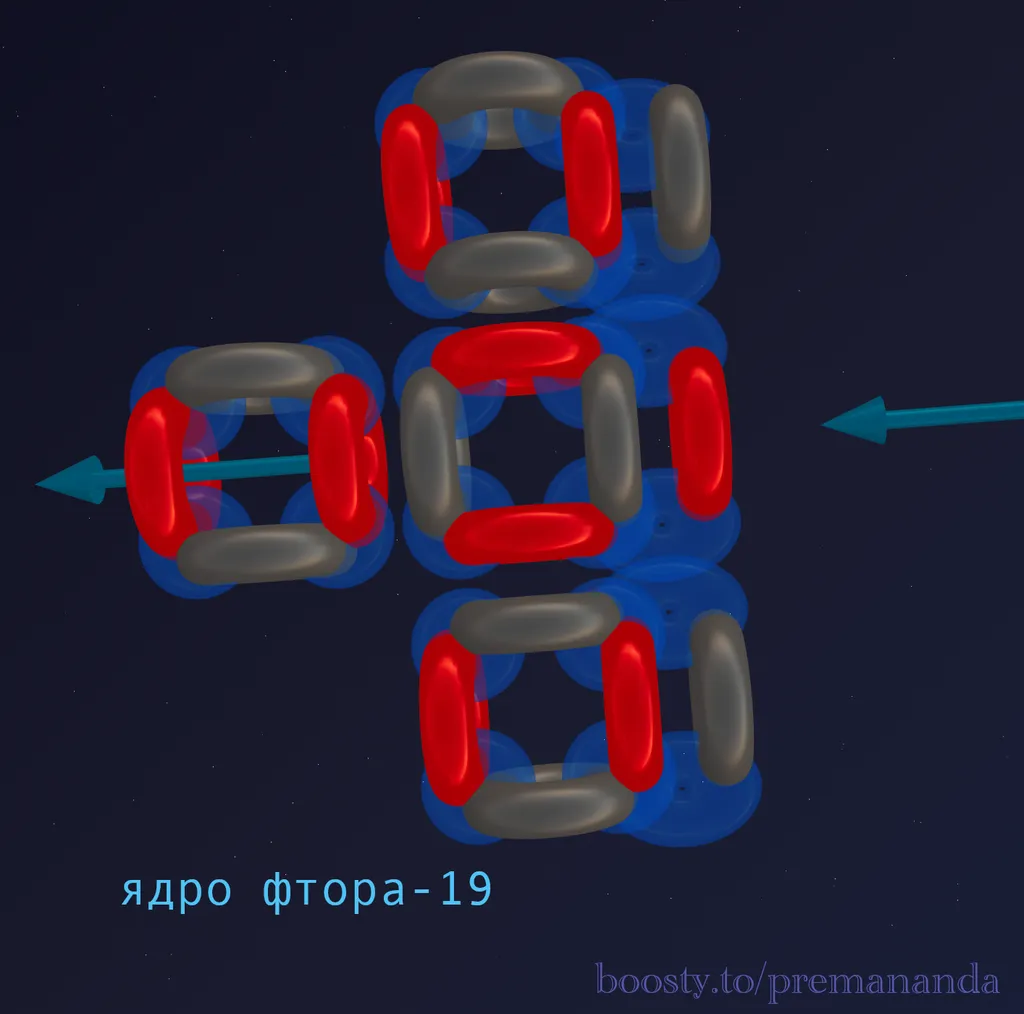
Fluorine-19 is the only stable isotope of fluorine.
Composition: 9 protons + 10 neutrons = 19 nucleons.
Division into blocks:
- 16 nucleons = 4 alpha particles (like Oxygen).
- Remainder: 3 nucleons = 1 proton + 2 neutrons (triton).
Formula: ¹⁹F = 4α + t
🔬 Building the Model
Step 1: Basic Oxygen Structure
Let’s start with what we already know. Oxygen-16 consists of 4 alpha particles in a T-shaped configuration: 3 alpha particles form a linear chain, and the 4th attaches to the top center.
Step 2: Adding the Triton (Tail)
A triton (1p + 2n) is added to this structure — an asymmetrical superstructure.
Key question: Where does this tail attach?
Hypothesis: The triton splits into a proton and two neutrons. The two neutrons are distributed on the sides of the central axis, while the proton attaches to the central funnel, reinforcing it.
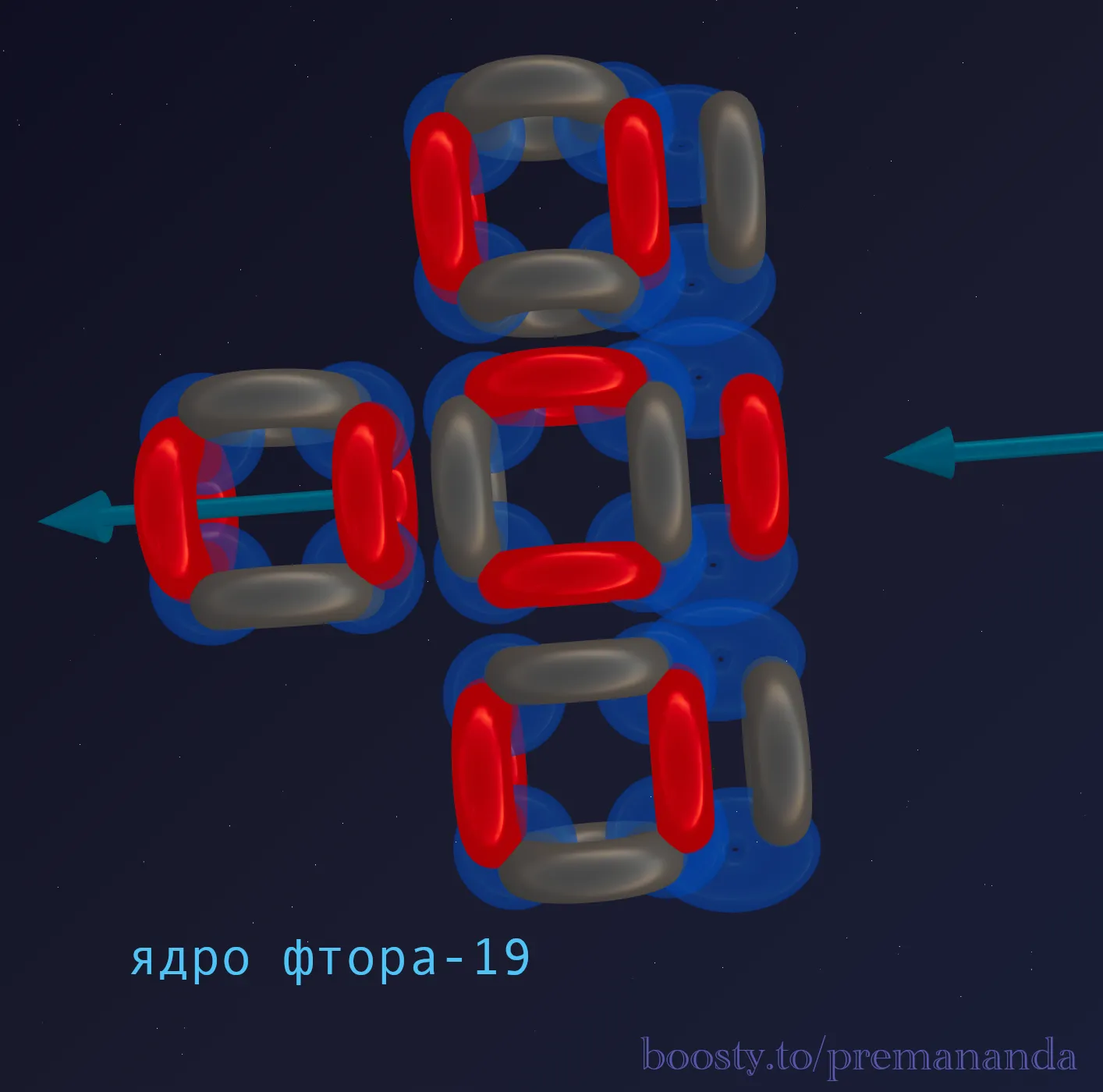
💀 Anatomy of a Predator: What do we see in the blueprint?
1. One Super-Powerful Funnel
Unlike Oxygen, where two funnels work symmetrically, in Fluorine, one active central funnel is reinforced by a proton, which most effectively sucks aether into the nucleus.
2. Excess Neutrons
10 neutrons to 9 protons is an unusual ratio for light elements. The excess neutrons create additional nucleon ties, but they also increase the internal pressure within the nucleus.
🔮 Model Forecasts and Reality
Forecast #1: Valency of 1
Looking at the model, we see that only one active funnel is available for full-fledged bond formation. The rest are significantly weaker. In reality: HF, CF₄, F₂ — in all compounds, Fluorine is strictly monovalent. ✓
Forecast #2: Record Electronegativity
The central proton “sitting” right inside the funnel turns the atom into a super-powerful vacuum cleaner. In reality: Fluorine is the absolute champion of electronegativity (4.0 on the Pauling scale). ✓
Forecast #3: Monstrous Reactivity
Fluorine doesn’t just “want” to form a bond — it needs to do so to stabilize its asymmetrical, highly tense structure. This explains why it reacts even with concrete and noble gases (like xenon). ✓
Forecast #4: Weak Bond in the F₂ Molecule
When two such “predators” meet, they start a tug-of-war for the aether. The bond turns out extremely tense and is easily broken. In reality: The F-F bond energy is anomalously low compared to other halogens. ✓

Forecast #5: Anomalously Strong HF Bond
Hydrogen is too weak to compete with Fluorine. It simply “gives up” its flow, and Fluorine completely dominates, creating an incredibly strong bond. In reality: The H-F bond is one of the strongest in chemistry. ✓
⚔️ Fluorine vs Oxygen: Comparing the Predators
| Parameter | Oxygen O (4α) | Fluorine F (4α + t) |
|---|---|---|
| Active funnels | 2 symmetrical | 1 reinforced |
| Structure | Stable | Tense (asymmetry) |
| Valency | 2 (rarely 3) | 1 (strictly) |
| Electronegativity | 3.5 (2nd place) | 4.0 (1st place) |
🧪 Nuclear Alchemy: Proof of Structure
If our hypothesis Fluorine = Oxygen + Triton is correct, nuclear physics should confirm it.
Reaction in stars:
¹⁹F + p → ¹⁶O + α
Engineering breakdown:
- We have Fluorine (Oxygen + Triton).
- A proton enters the nucleus.
- The proton completes the triton (1p + 2n) into a full-fledged alpha particle (2p + 2n).
- The alpha particle detaches and flies away.
- A pure Oxygen-16 framework remains.
This reaction acts as an excellent proof by contradiction, confirming our structural formula perfectly.
🌟 Summary
Fluorine is not just a “very active element.” It is the result of nature’s engineering design: an asymmetrical structure and a concentrated flow at a single point create the most effective chemical predator in the Universe.
Conclusion: We are not fitting facts to theory — we see the logic of nature through the architecture of its nuclei.
🔮 What’s next?
In the next part — Neon:
- A return to perfect symmetry.
- Why does a noble gas react with absolutely nothing?
- How to “seal” all funnels and create a chemical fortress.