Sulfur: The Alchemy of Oxygen. How Two Gases Become a Hard Yellow Stone. Part 19
“Architecture is frozen music.”
— Johann Wolfgang von Goethe
In the previous article we saw how Phosphorus (7α + t) became yet another element that disrupted symmetry in the period. The extra triton, latching onto Silicon’s rigid monolith, rotated one alpha particle and created a local zone of extreme overpressure — and the famous phosphorescent glow.
But Nature always strives for balance. The tension must be released, the triton must be completed, and the asymmetry must be eliminated.
Meet Sulfur — the element that brings us back to the perfect, symmetric mathematics of pure alpha blocks. Let’s see how this geometry elegantly solves one of chemistry’s great puzzles: variable valence (2, 4, and 6).
📐 Engineering Analysis of the Nucleus
Sulfur-32 is the primary stable isotope of Sulfur (95% in nature).
Composition: 16 protons + 16 neutrons = 32 nucleons.
Block decomposition:
- 32 nucleons = exactly 8 alpha particles (8 × 4 = 32);
- remainder: 0 — no incomplete fragments.
Formula: ³²S = 8α

We return to a clean architecture built exclusively from solid helium bricks. But true ideal symmetry is not here yet — Argon will be the symmetric one. Sulfur is an intermediate step: no triton “tail,” yet its 8α framework is still asymmetric in form.
🔬 Building the Model: The Third Column
Let’s trace the spatial evolution of the third period:
- Magnesium (6α) — base two-axis structure;
- Silicon (7α) — the second column added, a symmetric monolith with 4 funnels;
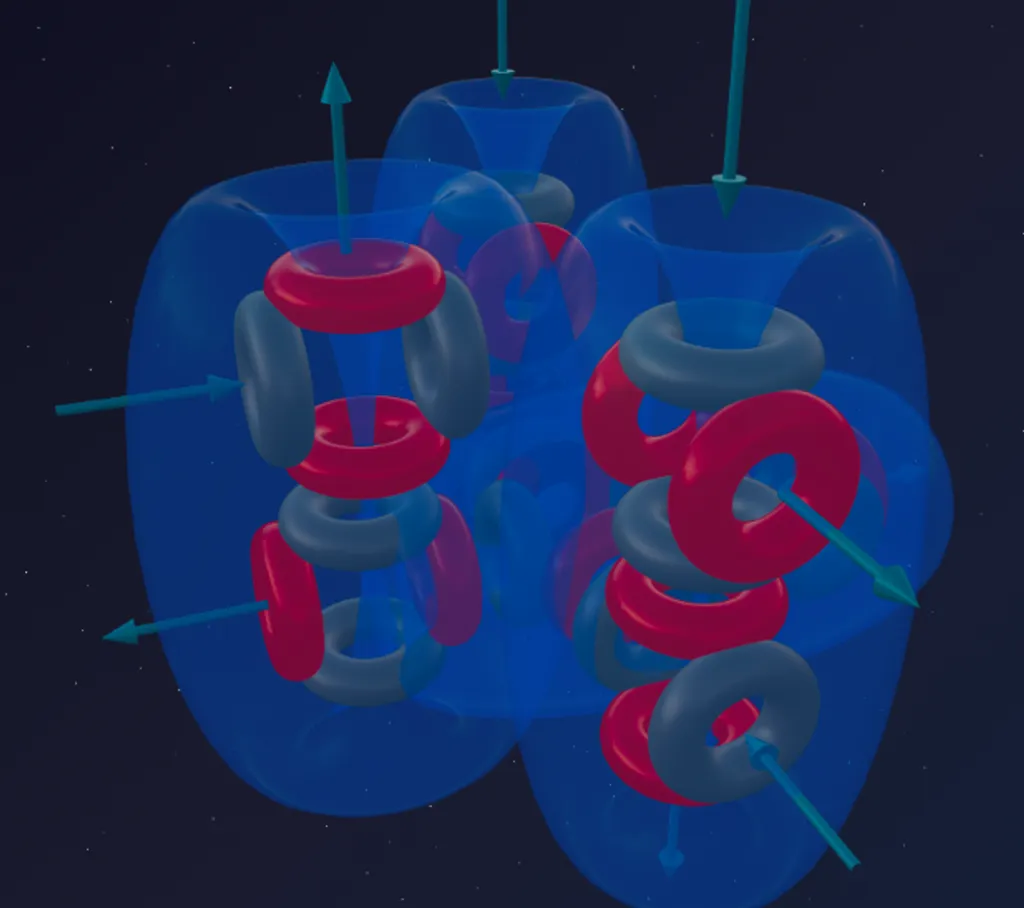
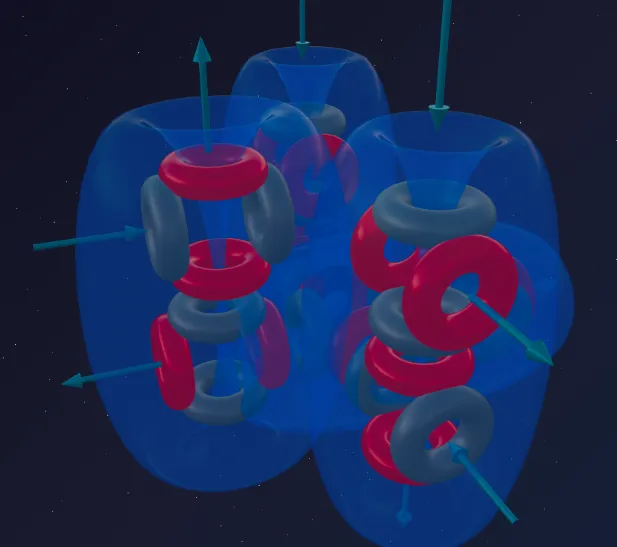
- Sulfur (8α) — the third column is added.
The eighth alpha particle attaches to the Silicon framework, opening new bonding planes. The structure becomes more massive, but not more symmetric — the internal pressure is distributed unevenly, and this determines Sulfur’s chemical behavior.
🏢 The Architecture of Funnels
Let’s trace the mathematical progression of open funnels:
- Neon (5α) — 0 funnels (sealed lock);
- Magnesium (6α) — 2 funnels (one axis opened);
- Silicon (7α) — 4 funnels (lateral columns activated);
- Sulfur (8α) — 6 funnels (third column activated).
The structure projects 6 active ports outward, divided into two groups: 4 less protruding (inherited from the Silicon base) and 2 more protruding (introduced by the 8th alpha particle).
🧪 Nuclear Alchemy: Proof of Structure
Nuclear reactions confirm the formula S = 8α.
Adding an alpha particle to Sulfur yields the next alpha-element — Argon:
³²S + α → ³⁶Ar + γ
Stripping an alpha particle returns us to Silicon:
³²S → ²⁸Si + α
A proton strike completes the triton into a full alpha particle, which flies off, exposing Phosphorus:
³²S + p → ³¹P + d
All reactions confirm the formula: S = 8α.
🔮 Model Predictions and Reality
Prediction №1: maximum valence 6
The number of outward-facing funnels determines the maximum number of chemical bonds. The 8α framework of Sulfur has exactly 6.
Reality:
- SF₆ (sulfur hexafluoride) — Sulfur bonds with exactly six Fluorine atoms — a perfect match with the model;
- H₂SO₄ (sulfuric acid) — Sulfur uses all 6 bonds to hold its oxygen environment — a perfect match.
Prediction №2: solving the variable valence puzzle (2, 4, and 6)
School chemistry simply makes students memorize: “Sulfur exhibits valences of 2, 4, and 6.” Why are the numbers even? Why do they change? Aether dynamics gives a geometric answer.
Depending on the strength of its partner, Sulfur activates its ports in stages:
- Valence 2 (H₂S — hydrogen sulfide): weak Hydrogen atoms latch only onto the two most outward-protruding funnels. The remaining four ports are not activated.
- Valence 4 (SO₂ — sulfur dioxide): powerful Oxygen activates the four less protruding ports. The two remaining funnels are closed onto themselves.
- Valence 6 (SO₃, SF₆): the most aggressive partners literally “throw open” the Sulfur atom, forcing all 6 ports into action.
Variable valence is the sequential activation of the functional nodes of a 3D structure. The even numbers arise from the symmetric, pairwise switching-on of funnels — a perfect match with the model.
Prediction №3: polymerization into “crowns” (S₈)
Sulfur has 6 funnels, yet it does not form an ultra-strong monolithic solid like diamond. The two most outward-protruding funnels are ideal for linear bonding, but due to the bulkiness of the atoms the chain bends — and on the eighth atom it closes back on itself.
The molecule S₈ forms — a molecular “crown.” This is why Sulfur is a solid, but a brittle one (crowns are easy to separate from each other) and low-melting (melts at 115°C) — a perfect match with the model.
Prediction №4: kinship with Oxygen
Sulfur sits directly below Oxygen in the periodic table. Their aether dynamics kinship is clear:
- Oxygen = 4α (2 funnels);
- Sulfur = 8α (6 funnels).
Where Oxygen forms oxides (H₂O, Fe₂O₃), Sulfur forms sulfides (H₂S, FeS₂ — pyrite). The heavy base of Sulfur (8α) makes its flow more inertial — Sulfur is a “slower” oxidizer than the light, aggressive Oxygen — a perfect match with the model.
🌟 Summary
Sulfur (8α) is the addition of the third column in the third period. No triton “tail,” but no ideal symmetry either — that will come only with Argon.
The aether dynamics of the 8α framework brilliantly predicts exactly 6 chemical funnels divided into two functional groups. This allows us to explain variable valence (2, 4, 6) without quantum paradoxes — as the step-by-step activation of different ports depending on the strength of the chemical partner.
🔮 What’s Next?
Symmetry is partially restored, but the period is not yet closed. In the next part — Chlorine:
- what happens when a triton is attached to Sulfur’s 8α framework;
- why Chlorine (8α + t) became one of the most aggressive predators on the planet;
- how one proton turns a yellow stone into a suffocating green gas.
🛠️ Build Your Own Model!
Try building the Sulfur-32 nucleus in the online constructor: