
Atomic Structure. Carbon and the Geometry of Life. Part 4
“Simplicity is the ultimate sophistication.”
— Leonardo da Vinci
In the previous parts, we established why helium-4 is the ideal “building block” of matter, and why nuclear stability requires not merely a sum of protons and neutrons, but the correct geometric arrangement of tori that ensures the closure of aether flows.
Today we turn to the most important element for life as we know it — carbon-12.
🧩 The Carbon Puzzle
Carbon-12 is composed of three alpha particles (6 protons, 6 neutrons).
The key question: if two alpha particles cannot hold together (as in beryllium-8), why do three suddenly form a stable structure?
One hypothesis is that they form a triangle. However, a closer look at aether dynamics makes it clear that the rigid “cross” geometry of alpha particles makes a triangular arrangement extremely difficult to achieve without flow conflicts.
The solution that fits our model perfectly is a linear structure with a 90-degree rotation.
📐 The Architecture of the Carbon Nucleus
Take a look at the model of the carbon-12 nucleus:

The structure resembles a chain of three links:
- the two outer alpha particles are oriented vertically;
- the central alpha particle is rotated 90 degrees relative to them (horizontally).
Why Is This Structure So Exceptionally Stable?
This is governed by a “tongue-and-groove” principle at the level of aether flows:
- Direct closure: the protons (“pumps”) of the central particle point directly into the neutrons (“intakes”) of the outer particles. The flow closes directly, without turbulence.
- Rigid axis: the central link literally “holds” the side links, forming a rigid axis.
- Attraction effect: according to Bernoulli’s principle, the high velocity of aether flows between nucleons creates a zone of reduced pressure that effectively “glues” the structure from within.
⚛️ The Nature of the Electron
Before discussing valence, we need to clarify what an electron is in our model. An electron is not a ball orbiting the nucleus, nor a probability cloud.
An electron is a flow of aether.
It is a vortex loop generated by the nucleus itself. A proton (acting as a high-pressure pump) expels a jet of aether that loops through space and returns. That loop is what we call an electron. The electron generation mechanism is discussed in detail in Part 1 of the series.
💡 Aether Dynamics Interpretation of Electron Shells
Mainstream science divides carbon’s electrons into two groups:
- Inner electrons (1s²): 2 electrons close to the nucleus, very tightly bound, and not involved in chemical reactions.
- Valence electrons (2s² 2p²): 4 electrons farther out, more loosely bound, and responsible for the element’s chemistry.
How does our linear model explain this?
Look at the chain of three alpha particles again. We have one central and two outer units.
Inner Shell (Analogue of 1s²)
The central alpha particle is “sandwiched” between its two neighbors. Its protons — exactly 2 of them — are engaged in holding the nuclear structure together. The aether flows from these protons are short, dense, and closed within the structure. They form the “indestructible core” that plays no role in chemistry.
Valence Shell (Analogue of 2s² 2p²)
The two outer alpha particles have open sides facing the outside world. Together, this gives 4 protons (2 on each end). Their aether flows are not blocked by neighbors — they extend far outward and form wide loops. These 4 flows are precisely the “valence electrons” with which carbon bonds to other atoms.
🔌 The Secret of Valence: 4 Ports for Life
Now it is clear where the valence of 4 comes from. It is not an abstract number — it is real geometry:
- Injection: 4 outer protons generate active vortices (valence electrons).
- Suction: of the 6 neutrons in the nucleus, 2 are engaged in the internal bond with the central particle, while the remaining 4 neutrons at the ends are free and act as “suction ports.”
These 4 suction zones and 4 injection zones together create carbon’s unique ability to build highly complex molecules.
Formation of Methane (CH₄)
Hydrogen is a proton wrapped in an electron-torus. When 4 hydrogen atoms approach carbon, they are inevitably drawn toward the low-pressure zones — the 4 free “funnels” at the ends of the chain.
The hydrogen electrons are “plugged into” these funnels and merge with carbon’s electrons into a shared, dynamic structure. This is how a methane molecule is born.
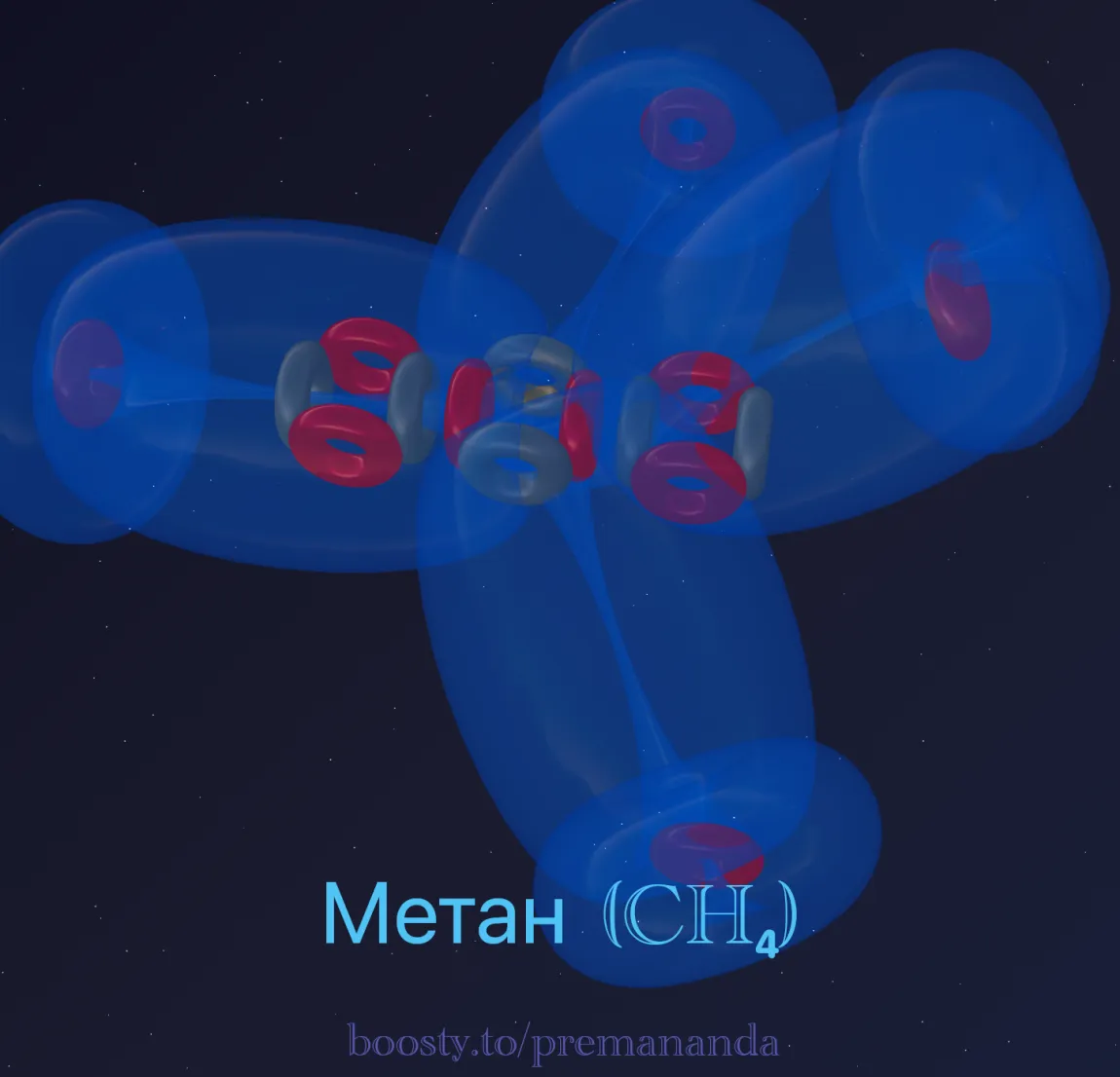
Most likely, the hydrogen nuclei sit at the corners of a regular tetrahedron, with the carbon nucleus at its center. This is driven by the system’s tendency toward minimum energy: since an electron in our model is a flexible vortex structure, the four electron loops naturally distribute themselves as evenly as possible in space, producing tetrahedral geometry.
🌟 Why Is Carbon the Basis of Life?
Carbon-12 is unique not because of any magic in its numbers, but because of its geometry:
- Chains: the linear structure allows it to form long chains — the backbone of organic chemistry.
- Branching: 4 valence ports enable the creation of branched structures.
- Stability: the symmetry of flows ensures stability when forming complex molecules.
- Versatility: it can bond with itself as well as with hydrogen, oxygen, and nitrogen.
Conclusion: life is based on carbon not by coincidence, but because its geometric structure is perfectly suited for building complex, stable, and functional molecules.
🔮 What’s Next?
In the next part, we will look at:
- the structure of nitrogen-14;
- where the valence of 3 comes from and what a “lone pair of electrons” is;
- why nitrogen forms triple bonds (N≡N);
- how nitrogen participates in the formation of amino acids — the building blocks of proteins.